GED Related Exams
GED-Science Exam



Catalysts are chemical substances that increase the rate of a chemical reaction but are not changed by the reaction. Enzymes are special catalysts produced in body cells and are composed of proteins.
Which statement is true?
In a population cycle, predators eat and reduce the number of prey. If the number of predators is reduced, the number of prey will gradually increase. Eventually, the predator population will increase, and the population cycle will continue.
Which pair best demonstrates a predator-prey relationship?
To begin a fission reaction, a neutron is shot at high speed into the nucleus of an atom of Uranium-235. This process releases a large amount of energy contained within the nucleus. The diagram above illustrates this process as well as possible by-products.
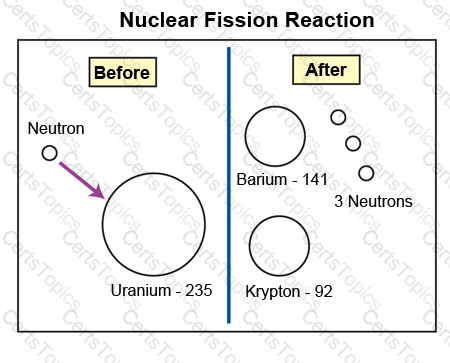
Based on the diagram and information, which statement best describes the fission reaction shown?